|
The cookie is used to store the user consent for the cookies in the category "Performance". This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Other. The cookies is used to store the user consent for the cookies in the category "Necessary". The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". The cookie is used to store the user consent for the cookies in the category "Analytics". These cookies ensure basic functionalities and security features of the website, anonymously. Necessary cookies are absolutely essential for the website to function properly. The particle model describes the energy, arrangement and movement of particles in solids, liquids and gases. This results in low numbers of particles in a given space. ▸ Are closely packed, in a free arrangement.Įxplanation: Particles have high energy and virtually no attraction between them therefore they are free to move apart. Particles in liquids: ▸ Have medium kinetic energy. ▸ Are closely packed, in a fixed arrangement. ▸ Are very strongly attracted to each other. Particles in solids: ▸ Have low kinetic energy. How are particles in solids attracted to each other? Evaporation is sometimes confused with boiling. Brownian motion is the random movement of fluid particles. There are energy changes when changes in state occur. The kinetic particle theory explains the properties of solids, liquids and gases. 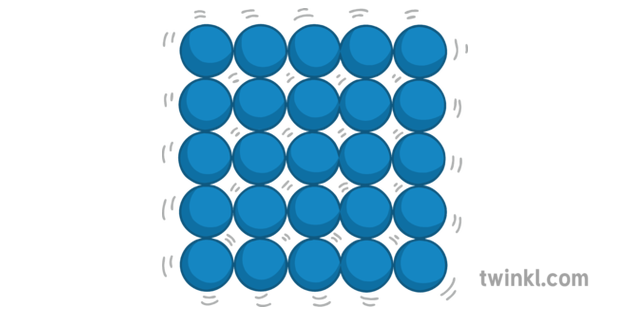
How does the kinetic particle theory explain state changes? When a solid is heated the particles gain energy and start to vibrate faster and faster. Even though they are vibrating this is not enough to disrupt the structure. In a solid the strong attractions between the particles hold them tightly packed together. If it is cooled the motion of the particles decreases as they lose energy. What happens to the motion of particles in a solid? Processes in which matter changes between solid and gaseous states are sublimation and deposition. Processes in which matter changes between liquid and gaseous states are vaporization, evaporation, and condensation. Which changes of state occur between solids and gases? The particles are held together too strongly to allow movement from place to place but the particles do vibrate about their position in the structure. In a solid, the particles pack together as tightly as possible in a neat and ordered arrangement. When thermal energy is added to a substance, its temperature increases, which can change its state from solid to liquid (melting), liquid to gas (vaporization), or solid to gas (sublimation). How does energy affect the state particles are in? Gas particles move about freely, so gases fill their containers. Liquid particles move more loosely, so they can take the shape of their containers. 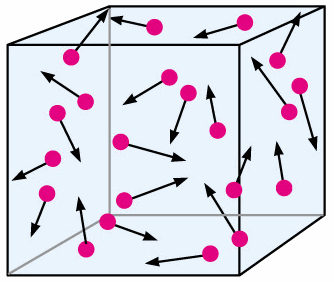
Particles in a solid vibrate in place, so solid substances are rigid. The particle motion determines the state of matter, and each state behaves differently. How does the movement of particles affect the state of matter? In the solid the particles vibrate in place. Their vibration is related to their kinetic energy. Solid � In a solid, the attractive forces keep the particles together tightly enough so that the particles do not move past each other. How does particle movement affect a solid? Further heating provides more energy until the particles start to break free of the structure. Initially the structure is gradually weakened which has the effect of expanding the solid. How does particle movement and energy affect a solid? 7 Why are particles free to move in space?.6 How does the kinetic particle theory explain state changes?.5 What happens to the motion of particles in a solid?.3 How does energy affect the state particles are in?.2 How does particle movement affect a solid?.1 How does particle movement and energy affect a solid?.The particles in the diagrams could be atoms, molecules or ions depending on the type of substance, eg ionic compounds, simple molecules, giant covalent structures, and metals. The model is used to explain the physical properties of solids, liquids and gases. 
The particle theory of matter is a model that describes the arrangement and movement of particles in a substance.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
